Aortic dissections are one of the most catastrophic and life-threatening events affecting the aorta; this condition brings tens of thousands of Americans to the emergency room each year.
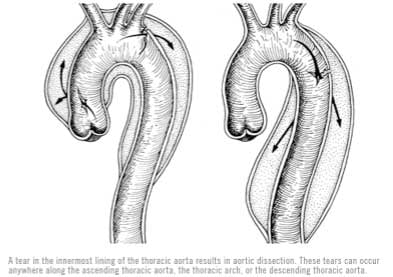
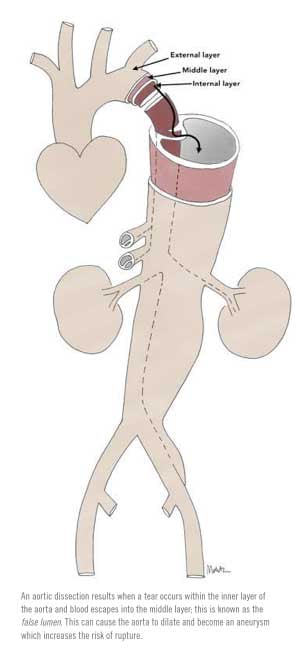
An aortic dissection results when a tear occurs within the inner layer of the aorta and blood escapes into the middle layer of the blood vessel. This false passage can travel along the length of the aorta and is known as the false lumen. Once the tear has begun, the high blood pressure will continue to perfuse the false lumen and expand this dissection forward and/or backward. Depending on the site of aortic tear, if it propagates backward, the damage can go on to involve the heart vessels and the valves, and if it propagates forward, the tear can affect other major branches of the aorta that perfuse the abdominal organs and lower extremities. As the false lumen expands, the blood supply through the true lumen can become choked off, resulting in organ malperfusion and complications including stroke, heart attack, congestive heart failure, kidney failure, paraplegia, and limb-threatening ischemia.
 The majority of patients are between the ages of 50-80 years, although younger patients with hypertension or connective tissue disease are also at risk. Patients describe the pain associated with acute aortic dissection as “sharp” and “tearing,” often located in the chest and between the shoulder blades and can mimic symptoms of a heart attack.
The majority of patients are between the ages of 50-80 years, although younger patients with hypertension or connective tissue disease are also at risk. Patients describe the pain associated with acute aortic dissection as “sharp” and “tearing,” often located in the chest and between the shoulder blades and can mimic symptoms of a heart attack.
Classifying Dissections
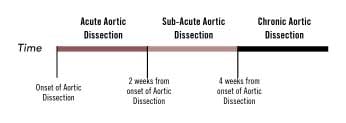
 In addition to aortic dissection, symptomatic conditions such as aortic intramural hematoma and aortic penetrating ulcer are generally classified as acute aortic syndromes and require immediate medical, endovascular, or surgical attention. Our understanding of aortic dissection has evolved over the past few decades; historically, treatment strategies were based on patient presentation and the timing of symptom onset. Dissections were classified as acute if symptoms occurred within 2 weeks, subacute for symptoms lasting between 2 and 6 weeks, and chronic if symptoms were present for more than 6 weeks. Recent evidence suggests that optimum treatment strategies should be based on not just the timing, but also the symptoms as well as the initial aortic morphologic findings noted on computed tomography (CT) scans.
In addition to aortic dissection, symptomatic conditions such as aortic intramural hematoma and aortic penetrating ulcer are generally classified as acute aortic syndromes and require immediate medical, endovascular, or surgical attention. Our understanding of aortic dissection has evolved over the past few decades; historically, treatment strategies were based on patient presentation and the timing of symptom onset. Dissections were classified as acute if symptoms occurred within 2 weeks, subacute for symptoms lasting between 2 and 6 weeks, and chronic if symptoms were present for more than 6 weeks. Recent evidence suggests that optimum treatment strategies should be based on not just the timing, but also the symptoms as well as the initial aortic morphologic findings noted on computed tomography (CT) scans.
Once aortic dissection is diagnosed, immediate medical treatment includes the use of anti-hypertensive drugs, vascular surgery, and possibly cardiac surgery consultation. The optimal management is directed toward identifying whether an aortic dissection is complicated or uncomplicated. Patients with complicated dissections present with organ malperfusion, refractory pain, or rupture, and they require immediate surgical or endovascular treatment. For the vast majority of patients in whom the aortic tear occurs in the descending thoracic aorta, endovascular repair is considered the “gold standard.” In patients with ascending thoracic involvement, surgical repair is preferred, albeit advances in techniques and technology will likely allow surgeons to treat many of the ascending thoracic aortic dissections by endovascular means as well.
For patients with uncomplicated dissections, the ideal mode of therapy—be it medical management or intervention—remains unclear. Our understanding is evolving, and this field is rapidly changing.
Endovascular Solutions
Endovascular repair of aortic dissections requires the surgeon to gain remote access to the aorta through femoral arteries in the groin. Under x-ray guidance, the surgeon/operator navigates wires, catheters, and stentgrafts within the arteries and places the stentgraft strategically in healthy aorta above and below the dissections, thereby excluding blood flow within the torn false lumen. Adjunctive endovascular and hybrid surgical procedures are often required to maintain vital organ perfusion and thrombosis of the dissected aortic false lumen. The operating theater needs to be well equipped and the staff well trained.
These procedures are generally considered to be some of the most complex and require a detailed analysis of imaging, case planning, procedure implementation, and postoperative recovery. After endovascular repair, patients require lifelong follow-up with routine clinical evaluation and appropriate imaging to assess the integrity of the stentgraft, as well as to monitor for any development of future dissections or aneurysms.
Predicting Outcomes
Research findings suggest that (1) persistent blood flow and patency of the false lumen are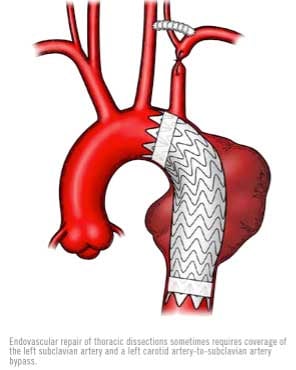 predictors of increased long-term patient mortality, (2) a false lumen diameter of > 22 mm is associated with increased future aneurysmal degeneration and mortality, (3) false lumen patency and maximum total aortic diameter of > 4 cm have the worst outcomes and the highest risk for future aneurysmal enlargement, (4) endovascular repair that leads to complete false lumen thrombosis has the best long-term prognosis, and (5) recurrent symptoms, thoracic aortic aneurysm dilatation of > 55 mm, or yearly increase of > 4 mm are predictors of worse outcomes and need repair.
predictors of increased long-term patient mortality, (2) a false lumen diameter of > 22 mm is associated with increased future aneurysmal degeneration and mortality, (3) false lumen patency and maximum total aortic diameter of > 4 cm have the worst outcomes and the highest risk for future aneurysmal enlargement, (4) endovascular repair that leads to complete false lumen thrombosis has the best long-term prognosis, and (5) recurrent symptoms, thoracic aortic aneurysm dilatation of > 55 mm, or yearly increase of > 4 mm are predictors of worse outcomes and need repair.
Minimally invasive endovascular treatment strategies for aortic dissections have transformed how we manage patients with complicated and uncomplicated aortic dissections, leading to significant reductions in patient morbidity and mortality. Prompt diagnosis and mobilization of appropriate surgical teams, however, remain critical for optimum patient outcomes.
To learn more about Acute Aortic Dissections, check out our other blogs.
This article is a V-AWARE Property. Center for Vascular Awareness, Inc. (www.myvascularhealth.org)








_2.jpg)